Custom, Simple, Fast
Steam sterilisation technology exposes your products with saturated steam under pressure. Steam enhances the ability of heat to kill microorganisms by reducing the time and temperature required to denature or coagulate proteins in the microorganisms. Steam sterilisation cycles generally have three phases including conditioning, exposure and exhaust.
According to the ISO 17665 standard
Steam sterilisation is a simple but very effective method of decontamination. Products are sterilised by being exposed to saturated steam at high temperatures (121°C to 134°C). The exposure time of the device to steam is between 3 and 15 minutes, depending on the heat generated.
For effective steam sterilisation, it is essential that the steam covers all surfaces of the device. To ensure optimal conditions, our autoclaves are equipped with built-in meters that display temperature and pressure conditions over time. Biological indicators and a colour indicator are also used to assess the performance of the autoclave.
Medistri is qualified to perform steam sterilisation in accordance with ISO 17665, ANSI and AAMI.
ISO 17665 specifies the various requirements for the development, validation and routine control of a steam sterilisation process for medical devices. The procedures that are covered by ISO 17665-1:2006 for steam sterilization include, but are not limited to
- Saturated steam exhaust systems
- Saturated air/steam exhaust systems
- Air/steam mixtures
- Water vaporization
- Water immersion
Our team prepares the validation protocol, performs all the required sterilisation cycles, carries out all the tests to generate a complete final report.
Our steam sterilisation services are not certified for the sterilisation of medicinal products for human and/or veterinary use.
Advantages
Disadvantages
A customer-focused approach to Steam Sterilisation
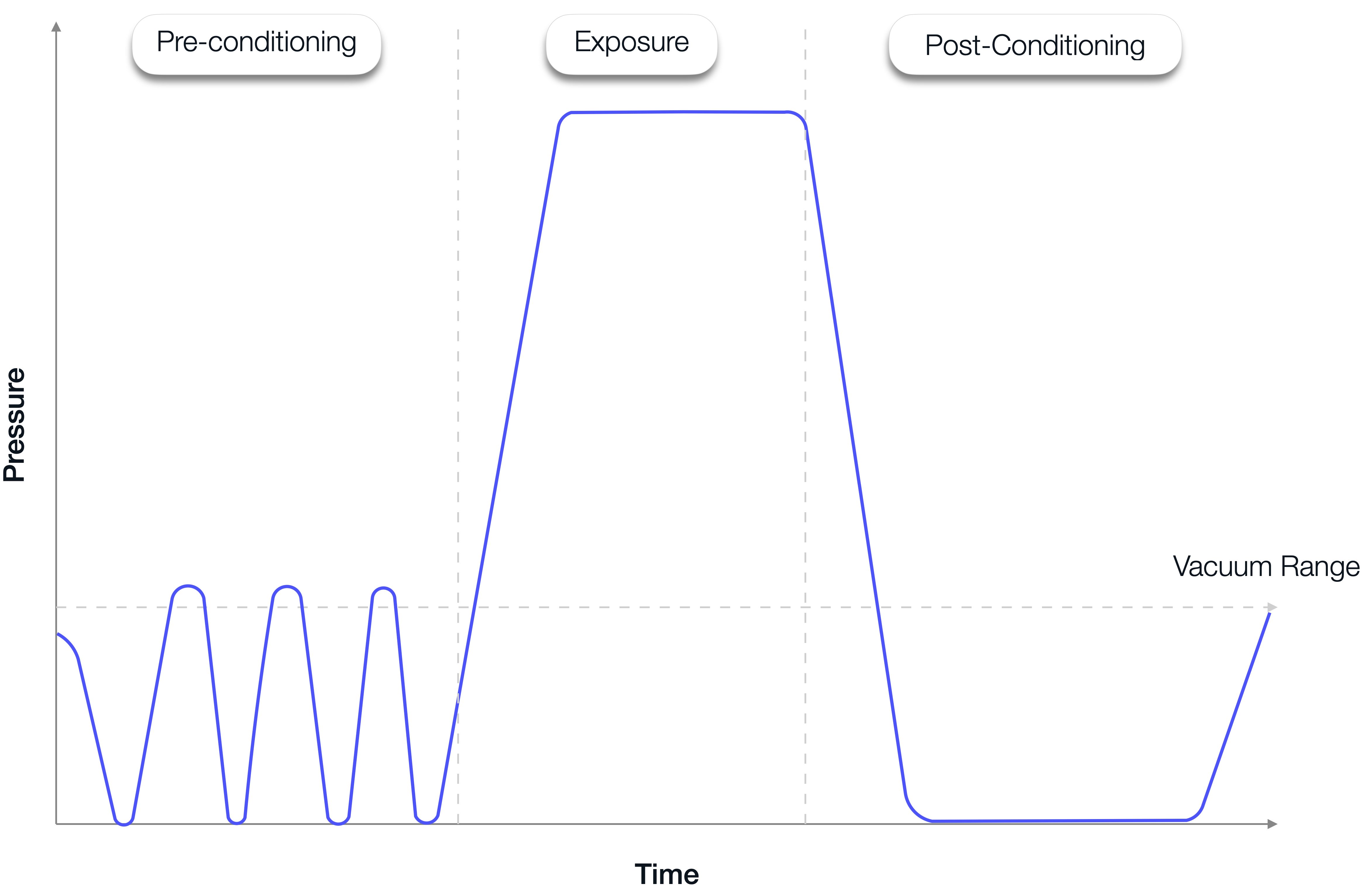
The 3 phases of a standard Steam Sterilisation cycle:
- Pre-Conditioning: During this phase, the air is removed from the chamber and the load is humidified by means of alternating vacuum and pressure pulses.
- Exposure: During this phase, the chamber temperature is raised to and held at the pre-validated sterilising temperatures for the per-validated exposure duration.
- Post-Conditioning: During this phase, dry loads are cooled and dried or liquid loads are cooled. The chamber is brought to atmospheric levels.
Learn more about the innovative healthcare companies using Medistri's Steam Sterilisation Services ➝
Packaging Validation Testing
Packaging Validation Testing is a series of multiple tests to help you design and validate your final medical or pharmaceutical packaging. This series of tests is performed in order to improve the numerous transportation risks involved and to improve patient health.
Medistri can help you perform the full range of transport simulation tests required under ASTM 4169 or ISTA series, to ensure final compliance with the requirements of ISO-11607 and help you deliver your vision.
Sterilisation for Pharmaceutical Vials
Learn how our customers sterilise their ready-to-use pharmaceutical vials at industrial scale 24/7 while ensuring the integrity and durability of their products.
Our Quality Certificates
Explore our list quality certificates and discover why the world's most innovative healthcare companies choose to work with Medistri.
Are you ready to get started?
Contact us and our qualified team will respond.