NEW
Product Packaging Solutions
Before a medical device reaches industrial production, development activities require packaging configurations capable of supporting product protection, transportation, sterilization evaluations, laboratory testing, and clinical preparation.
NEW
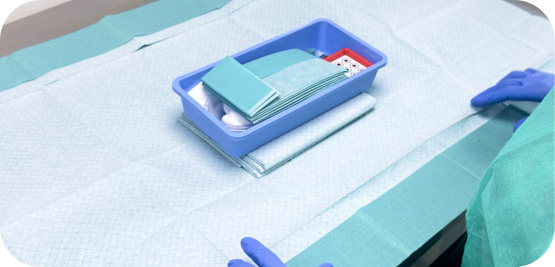
Medical Device Kit Packaging
Medical device manufacturers frequently manage products composed of multiple components that must be grouped, packaged, identified, and delivered as a single finished system. Whether intended for surgical procedures, hospital use, clinical evaluations, or commercial distribution, kit packaging plays a central role in ensuring that products are prepared according to their intended use, regulatory requirements, and distribution constraints.
Sterilization Validation Strategies for Medical Device Manufacturers
For medical devices and pharmaceutical products marketed as sterile, sterilization validation represents a critical transition point between development and routine commercial manufacturing. At this stage, manufacturers must demonstrate that the finalized product, packaging system, and manufacturing configuration can be sterilized under controlled and reproducible conditions while maintaining product performance and compliance with regulatory requirements.
Medistri Officially Listed Within the Swiss GLP Compliance Monitoring Program
Medistri is proud to announce that our laboratory is now officially listed within the Swiss GLP Compliance Monitoring Program under the oversight of the Swiss Federal Authorities and Swissmedic.
Sterile Barrier Integrity Testing for Medical Device Systems
Sterile barrier systems are designed to maintain the sterility of medical devices throughout storage, transport, and handling until the point of use. The performance of the sterile barrier system directly influences product safety, packaging reliability, and the ability to present the device aseptically within the clinical environment.
Environmental Monitoring for Medical Device Manufacturers
Medical devices are manufactured, assembled, and packaged in environments where contamination, whether particulate, microbiological, or chemical, can directly compromise product safety. Environmental monitoring is the systematic process of measuring and documenting the conditions in these environments, providing manufacturers with the evidence they need to demonstrate that their production areas are under control.
Post-Validation Training
Sterilization validation does not end with cycle approval. As regulatory expectations continue to evolve, maintaining validated conditions, controlling changes, and ensuring consistent routine release require structured expertise within internal teams.
An Overview of the "Big Three": Cytotoxicity, Irritation & Sensitization Testing
For nearly all medical devices, three biological endpoints form the foundation of a biological safety evaluation under ISO 10993-1: cytotoxicity, irritation, and sensitization. Commonly referred to as the “Big Three,” these endpoints are applicable across virtually all device categories, regardless of the nature or duration of patient contact. At Medistri, manufacturers are supported across these three endpoints through a combination of in-house in vitro testing and coordinated in vivo studies, ensuring that biological safety is demonstrated with scientific rigor and aligned with regulatory expectations.
Medistri Joins the Swiss GLP Compliance Monitoring Programme
At Medistri, we are proud to announce that our laboratory has officially been enrolled in the Swiss Good Laboratory Practice (GLP) Compliance Monitoring Programme. The formal decision was issued by the Notification Authority for Chemicals (NAChem) at the Federal Office of Public Health, following a successful inspection process conducted by Swissmedic. Our facility will appear on the official GLP register effective 13 May 2026.
Reprocessing and Cleaning Validation for Medical Devices
In healthcare manufacturing and clinical environments, contamination control is a fundamental regulatory and patient safety obligation. Medical devices may be exposed to biological contaminants through clinical use, or to chemical and particulate residues generated during manufacturing. Both reprocessing validation and cleaning validation are therefore required disciplines, each governed by distinct regulatory frameworks, yet sharing a common objective: demonstrating that contamination is effectively and reproducibly reduced to levels that do not compromise device safety, functionality, or intended performance.